 While valence bond theory is suitable for predicting the geometry and bond angle of HĢO, its prediction of electronic states does not agree with the experimentally measured reality. The difference between the predicted bond angle and the measured bond angle is traditionally explained by the electron repulsion of the two lone pairs occupying two sp 3 hybridized orbitals. This is in open agreement with the true bond angle of 104.45°. 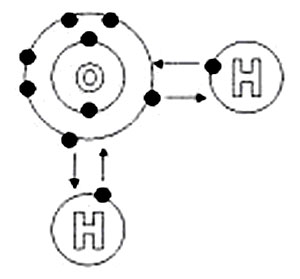
As such, the predicted shape and bond angle of sp 3 hybridization is tetrahedral and 109.5°. Valence bond theory suggests that HĢO is sp 3 hybridized in which the 2s atomic orbital and the three 2p orbitals of oxygen are hybridized to form four new hybridized orbitals which then participate in bonding by overlapping with the hydrogen 1s orbitals. Sp3 hybridization of H 2O Photoelectronic spectrum of H 2O Lewis structure and valence bond theory ĢO describes the bonds as two sigma bonds between the central oxygen atom and the two peripheral hydrogen atoms with oxygen having two lone pairs of electrons. Instead, several traditional and advanced bonding models such as simple Lewis and VSEPR structure, valence bond theory, molecular orbital theory, isovalent hybridization, and Bent's rule are discussed below to provide a comprehensive bonding model for HĢO, explaining and rationalizing the various electronic and physical properties and features manifested by its peculiar bonding arrangements. 
Despite being one of the simplest triatomic molecules, its chemical bonding scheme is nonetheless complex as many of its bonding properties such as bond angle, ionization energy, and electronic state energy cannot be explained by one unified bonding model. Lewis Structure of H 2O indicating bond angle and bond lengthĢO) is a simple triatomic bent molecule with C 2v molecular symmetry and bond angle of 104.5° between the central oxygen atom and the hydrogen atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
